 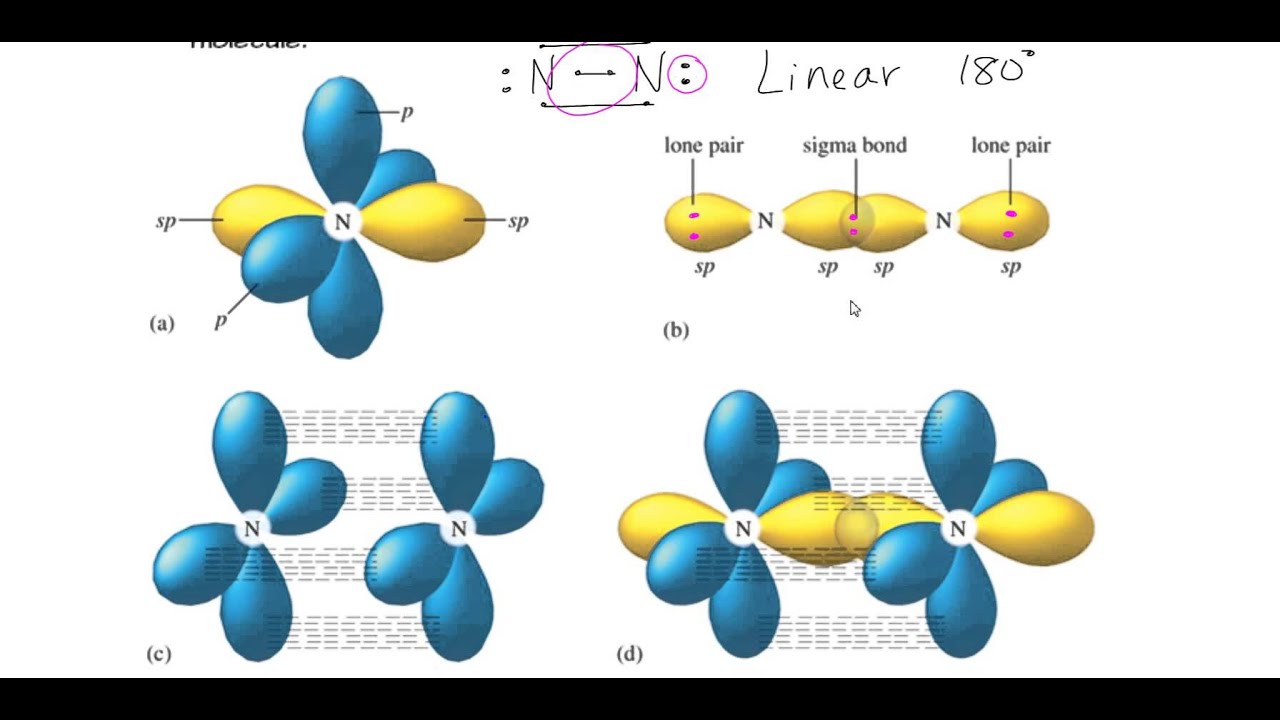
If you would use that formula for propane, number of sigma bonds will be 3 + 8 - 1 = 10, as shown above. Coming to the doubt about sigma and pi bonds, remember that the first bond between 2 atoms is always a sigma bond and any additional bond between these 2 atoms will be a pi bond. You can find sp2 bonding when carbon has a ONE. Number of sigma bonds = number of atoms - 1 (which you can determine using the molecular formula ) and then then the value of the steric number gives us the number of hybridized orbitals. For a carbon with 1 double bond and 2 single bonds, the orbitals will become 33 's' and 66.7 'p' making it 'sp2.' If there is a triple bond and a single bond, the orbitals will adjust again to become 50 's' and 50 'p.' So to summarize - You can find sp3 bonding when a carbon has 4 single bonds. metallic bonding, delocalization, sigma & pi bonds, bond length & energy. Draw the total number of sigma and pi bonds of the sulfur trioxide molecule, SO 3. 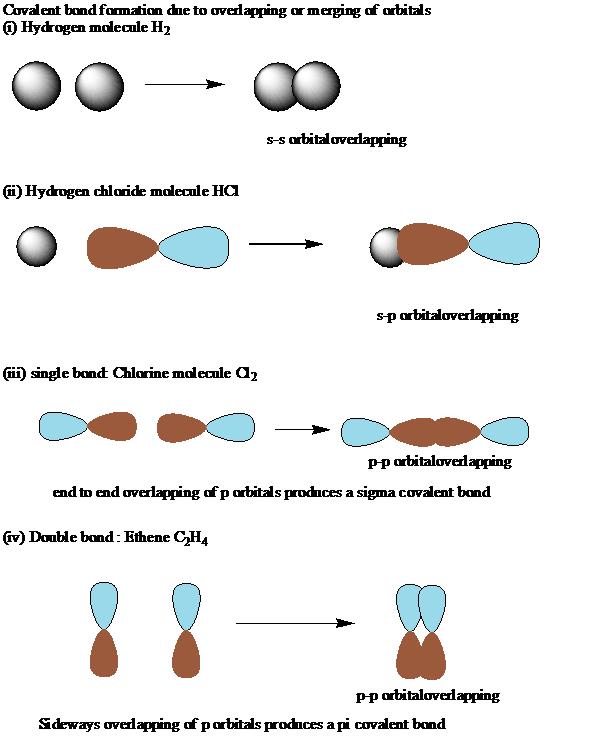
Also, in this particular case ( for saturated aliphatic alkanes ) you can use a formula: B, 8 more Short Answer Localized Electron Bonding: Properties of Molecular. It is a saturated compound, so you will only have simple ( sigma ) covalent bonds - there are no pi bonds. What are the Sigma and Pi bonds Sigma bond It is the only bond in the single covalent bonds, and it is symbolized by the Greek letter sigma (). Let's take the propane extended structure formula (which i stole from the answer above): Each simple covalent bond (represented by the line between 2 atoms) will be a sigma bond Īny other different from that ( double, triple covalent bonds ) will still have one sigma bond, the rest of the lines being counted as pi bonds Sigma bonds are formed by end-to-end overlapping and Pi bonds are when the lobe of one atomic orbital overlaps another. Start off by writing down the respective compund using the extended structure model. Sigma and pi bonds are formed by the overlap of atomic orbitals. Here you have a more in-depth analysis of structure formulas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
